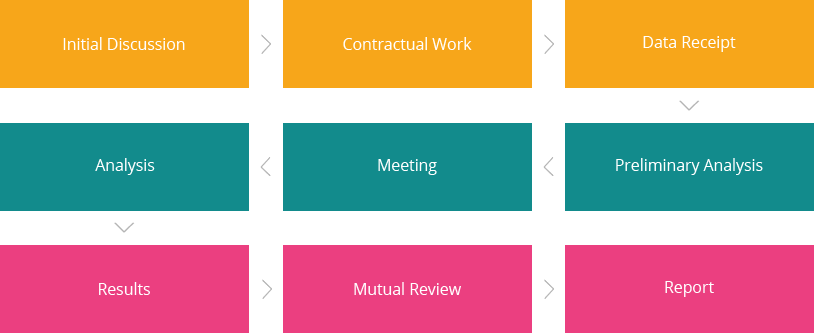
About service
C&R Research provides an one-stop drug development process through its integrated services including new drug model design and overall clinical development services, under mutual collaboration with Q-fitter, Korea’s first and only commercial pharmacometrics service provider. With vast experience in pharmacometrics and clinical pharmacology, Q-fitter’s experts offer high quality services all phases of clinical trials.
Phase 0 – Phase IIA
- Early-Phase Trial Designing
- Protocol Development
- Trial-Related Document Development: CRF (Case Report Form), ICF (Informed Consent Form), Various Forms, etc.
- Patient-Based Early-Phase Trial Research Team Construction
- New Therapeutics Regulatory Affairs: Cell Therapy, Gene Therapy, etc.
- Clinical Trial Participation as an Investigator
- CRF Fill-Up + QC (Quality Control) /QA (Quality Assurance) Activities)
- Statistical Analysis and CSR (Case Study Report) Writing
Pre-clinical ~ PoC
- Population PK-PD Analysis
- Nonclinical PK/PD Modeling & Simulation
- First-in-Human (FIH) Dose Estimation Based on Nonclinical PK/PD Modeling
- Human PK/PD Evaluation via Early Clinical Trial Data
- Non-Compartmental Analysis (NCA) on Early Clinical Trial Data
- Drug Development Consultation on Clinical Study Design
- Pharmacometric Consultation: Liaison services with bioanalytical facilities and sponsors to ensure PK data integrity and to provide data interpretation
- Scientific Report Writing
One-stop, quick-win center
To accelerate the drug development in all phases, Q-fitter offers one-stop drug development service by combining vast experience working across early clinical trials and pharmacometric analysis with C&R Research’s clinical trial service know-how. Q-fitter’s experts provide rapid and convenient consulting for drug development and clinical trial solution through analysis of preclinical data, clinical trial design and operation, and modeling & simulation.
Smooth communication-based services meeting clients’ needs
Q-fitter provide services customized to the needs of clients through all phases of clinical trials. Using modelling-simulation techniques, Q-fitter strives to improve the recently emphasized efficiency and productivity in the drug development process, coupled with the services that are suited to clients’ intended purpose (internal decision-making, design of the next-phase clinical trial, preparatory document generation for data submission to domestic and/or overseas regulatory authorities, and maximization of value-add effects in licensing-out of new drugs).
World-class clinical research professionals
Q-fitter’s professionals (principal investigator, clinical research coordinator, clinical pharmacologist) have at least 5 years of field experience in related clinical sector. They provide highly effective and productive services in early clinical trials, along with commercialized high quality pharmacometric services for the first time in Korea.
For more information